Difference between revisions of "Starch Conversion"
(→Gelatinization) |
(→Gelatinization) |
||
| Line 14: | Line 14: | ||
Gelatinization is a process that requires free water for the swelling and breaking the hydrogen bonds that hold the crystalline structures in place. If free water is limited due to a high concentration of starch (e.g. overly thick mash conditions) less swelling takes place and a melting of the crystalline section needs to occur [Donald, 2004]. This leads to an increase in the gelatinization temperature. This limitation of free water can also be caused by the presence of sugars other dissolved solids. For corn starch it has been shown that a 25% sucrose solution increase the gelatinization temperature from 70 to 78C [Donald, 2004]. This could have been one of the factors why thick mashes showed a lower efficiency compared to thin mashes in the [[Effects_of_mash_parameters_on_fermentability_and_efficiency_in_single_infusion_mashing|Mashing Experiments]]. | Gelatinization is a process that requires free water for the swelling and breaking the hydrogen bonds that hold the crystalline structures in place. If free water is limited due to a high concentration of starch (e.g. overly thick mash conditions) less swelling takes place and a melting of the crystalline section needs to occur [Donald, 2004]. This leads to an increase in the gelatinization temperature. This limitation of free water can also be caused by the presence of sugars other dissolved solids. For corn starch it has been shown that a 25% sucrose solution increase the gelatinization temperature from 70 to 78C [Donald, 2004]. This could have been one of the factors why thick mashes showed a lower efficiency compared to thin mashes in the [[Effects_of_mash_parameters_on_fermentability_and_efficiency_in_single_infusion_mashing|Mashing Experiments]]. | ||
| + | |||
| + | As starch starts to gelatenize the viscosity of the liquid will increase. This can be noticed in brewing to some extend but by far less that what is commonly seen in cooking. The reason for that is the presence of enzymes (in particular α-amylase) that will start breaking down the amylose and amylopectin molecules as soon as they become accessible. This process reduces the viscosity of the mash and is therefore called liquification [Kunze, 2007]. Liquifying decoctions through a rest at 70-74C before further heating to a boil for example mitigates the risk of scorching the mash during the decoction boil. | ||
| + | |||
Revision as of 04:24, 10 January 2009
96% (?) of the solids in brewing wort are carbohydrates. Proteins make up only 3% and the rest are β-glucans, vitamins and minerals. Of the carbohydrates more than 95% are products of the starch conversion that happens in the mash tun. As a result the starch degradation is the main purpose of mashing. It produces fuel that the yeast needs for fermentation from the insoluble starch found in malt and mash tun adjuncts. The extend to which this conversion happens will determine the strength (gravity or extract content) of the produced wort and its fermentability as well as other quality parameteres.
In mashing starch conversion is preceded by gelatinization of the starches. While this is not necessary for conversion, as plants do exactly this when they germinate, it greatly speeds up the process though the exposure of a lot of substrate (amylose and amylopectin) from the starch granule to the starch converting enzymes (mainly α- and β-amylase).
Gelatinization
Starch granules are insoluble in cold water and will absorb only little water. The form a suspension that quickly settles once agitation stops. As the water is heated (50+ C) more and more water is absorbed and the granules start to swell [Narziss, 2005]. The water absorbed during this process can be up to 30 times the weight of the starch granule. This uptake of water initially happens within the amorphous growth rings. As a result the crystalline layers break open and separate from the starch granule as gelatenous sheets. At this point the crystalline structure is lost and the process becomes irreversible with respect to the shape of the starch granule [Shetty, 2006]. The starch granule has gelatinized.
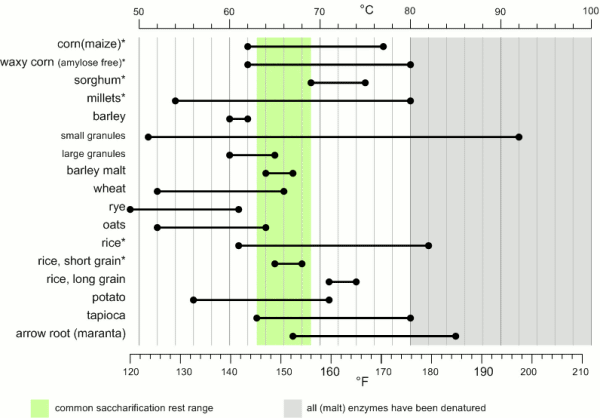
While the temperature range during with gelatinization occurs has been found to be quite narrow (~ 1C) for individual starch granules the temperature range between the gelatinization of the first granules and the complete gelatinization of all granules can be quite large. Figure 1 shows the temperature ranges for gelatinization for a number of starches. It can be seen that not all these starches fully gelatinize at temperatures that are encountered in a saccharification rest. In this case they will have to be gelatenized before that rest though either a cereal mash or the use of pre-gelatinized (e.g. flaked) forms. Another interesting aspect is the different gelatinization temperature ranges that have been determined for large and small barley granules. 90% of the starch in barley are large granules which will be gelatenized at saccharification rest temperatures while the rest are small starch granules which may not fully gelatenize until higher temperatures are reached. This effect can explain some of the efficiency benefits that can be gained from a mash-out or a decoction mash.
Gelatinization is a process that requires free water for the swelling and breaking the hydrogen bonds that hold the crystalline structures in place. If free water is limited due to a high concentration of starch (e.g. overly thick mash conditions) less swelling takes place and a melting of the crystalline section needs to occur [Donald, 2004]. This leads to an increase in the gelatinization temperature. This limitation of free water can also be caused by the presence of sugars other dissolved solids. For corn starch it has been shown that a 25% sucrose solution increase the gelatinization temperature from 70 to 78C [Donald, 2004]. This could have been one of the factors why thick mashes showed a lower efficiency compared to thin mashes in the Mashing Experiments.
As starch starts to gelatenize the viscosity of the liquid will increase. This can be noticed in brewing to some extend but by far less that what is commonly seen in cooking. The reason for that is the presence of enzymes (in particular α-amylase) that will start breaking down the amylose and amylopectin molecules as soon as they become accessible. This process reduces the viscosity of the mash and is therefore called liquification [Kunze, 2007]. Liquifying decoctions through a rest at 70-74C before further heating to a boil for example mitigates the risk of scorching the mash during the decoction boil.
[Shetty, 2006]:
- gelatinization: heating in excess water, endothermic reaction, loss of crystalline order
- irreversible w/r to the shape of the granule
- amylopecting and amylose may form new structures when cooled down -> retrogradation
- during heating -> swelling to up to 30x its weight. reversible process
- retrogradation is cause for hard stale bread
[Donald, 2004]
- irreversible order -> disorder transition called gelatinization.
- swell, absorb water, leach amylose, loose crystallinity.
- birefringence is lost
- range is quite large for all granules 56-64 for wheat and potato but much smaller (1 C) for individual granules
- substantial swelling
- inital water absorption in the amorphous growth rings
- swelling driven process
- crystalline regions break open b/c of the swelling
- if water is limited (high density or presence of sugars) swelling doesn't take place as well and a melting of the crystalline sections needs to happen
- gelatinization temperature increases
- Maize starch: from 70C to 78C when placed in 25% sugar solution.
- sugar removes free water
[Briggs, 2004]
- sugar concentration raises gelatinization temperature
- enzymatic breakdown should happen before retrogradation occurs
- some starches require cooking temps between 90 and 120C (194-248F) before amylose lipid complexes are broken -> one of the reasons why cooking of some adjuncts is necessary.
- gelatinization temperatures of various starches (page 38), create table
[Narziss, 2005]
- starch is insoluble in cold water
- granules start to swell (50C +)
- then they get small fissures
- amylose leaches out
- amylopectin separates as gelatenous sheet
- viscous and sticky mass is formed\
- doesn't happen in mashing b/c of the enzymes
- but may cause problems in cereal cookers (especially rice)
[Kunze, 2007]
- 4 phases: ungelatenized, gelatinization, liquification, saccharification
- gelatinization temp dependent on growing conditions and crop year : 59 - 65C
[Kessler, 2008]
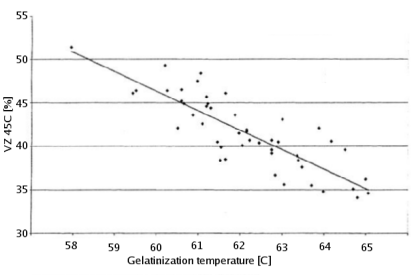
- VZ45 shows some correlation to gelatinization temperature
- gelatinization
- active enzymes
- emzyme temperature and pH optima
- mash parameters affecting conversion
References
- [Valclavik] Vickie A. Valclavik, Elizabeth W. Christian, Essentials of Food Science, Third Edition, Springer
- [Champe] Pamela C, Champe, Richard A. Harvey, Denise R. Ferrier, Biochemistry, Lippincott's Illustrated Reviews
- [Kunze, 2007] Wolfgang Kunze, Technologie Brauer und Maelzer, 9. Auflage, VLB Berlin
- [Narziss, 2005] Prof. Dr. agr. Ludwig Narziss, Prof. Dr.-Ing. habil. Werner Back, Abriss der Bierbrauerei, Technische Universitaet Muenchen (Fakultaet fuer Brauwesen, Weihenstephan). WILEY-VCH Verlags GmbH Weinheim Germany, 2005
- [Donald, 2004] A. M. Donald, Understanding Starch Stucture and Functionality, Chapter 5 in Starch in Food: Structure, Function and Applications By Ann-Charlotte Eliasson, CRC Press, 2004
- [Shetty, 2006] Kalidas Shetty, Food Biotechnology, CRC Press, 2006
- [Kessler, 2008] Dr.-Ing. Matthias Kessler, Dr.-Ing. Stefan Kreisz. Dipl.-Ing. Martin Zarnkov. Univ.-Prof. Dr.-Ing. Werner Back, Do Brewers need a starch modification index?, Brauwelt International, 2008/1