I’m starting to hit a stride with respect to yeast growth experiments. Last week I posted data on Access to Air and its Effect on Yeast Growth in Starters, this week I have data on the effect of the stir speed.
Some home brewers believe that as long as the starter is moving there is sufficient opportunity for the head space oxygen to diffuse into the starter beer and become available to the yeast as a nutrient. I never really believed that. Last week’s experiment showed that yeast growth does benefit from increased access to oxygen. So, if yeast growth is a reflection of the amount of oxygen taken up by the starter, then the impact of stir speed on oxygen uptake can be shown by its effect on yeast growth.
The setup of the experiment was simple. About 1200 ml of 8.8 Plato wort were prepared from DME and water. There is nothing magical about 8.8 Plato. That’s just how it came out after evaporation and topping off more water than planned. That wort was inoculated with WY2042, the low flocculant lager yeast that has become my favorite lab rat, to an initial density of 0.11 B/g or 10 B/L. That’s a pretty low initial cell density but I wanted the cell growth to be dominated by the available nutrients and not the health or reserves of the initial yeast population. The yeast was about 2 days old. That means 2 days after having been propagated last.
The inoculated wort was distributed among four 500 ml Erlenmeyer flasks. Not all flasks received the same amount of wort since I wanted to prevent the slow speed one from developing a vortex. After that the flasks were placed on identical PWM (pulse width modulation) controlled identical stir plates at about 22 C. The stir speed was adjusted to:
- low: no vortex, but yeast is kept in suspension
- medium: vortex reaches all the way to the stir bar
- high: strong vortex and lots of air bubbles are visible in the starter
I also added one experiment where I added one drop of Fermcap S to suppress foam formation. But the stir speed was so high that there was still foam formation even with the Fermcap S. This was also done on a high speed setting. No information about the actual RPMs of the speed settings is available.
All four starters were done uncovered in order to eliminate differences in gas exchange that may have resulted from crimping the aluminum foil differently.
The following are images that illustrate how the starters looked at the beginning:
 Slow speed setting. A mere dimple was noticeable on the surface of the starter. Total wort weight was 318 g. |
 Medium speed setting. A vortex was drawn all the way to the stir bar and created some foaming. Initial wort weight was 296 g. |
 Fast stir speed: bubbles were constantly drawn into the starter giving it a milky appearance. Initial wort weight: 256 g |
 Fast stir speed + Fermcap S. Same appearance as the “fast stir speed experiment”. Initial wort weight: 263 g |
Results and Discussion
This experiment did show a strong correlation between stir speed and yeast growth. The slower the stir speed was the less yeast was grown per initial gram of extract.
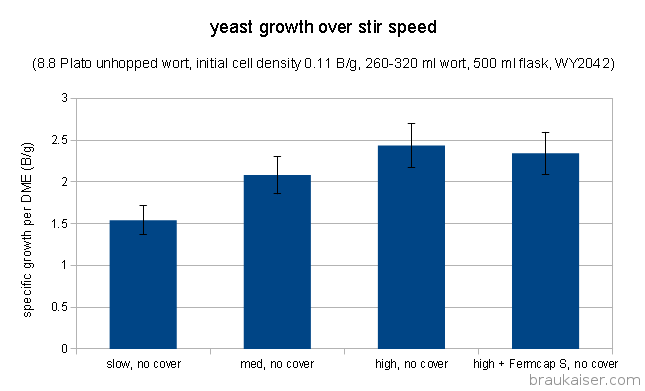
This is not surprising given the fact that a well developed vortex allows for a better gas exchange between head space and the starter. The growth rate of 2.08 B/g for the medium stir speed is in line with the growth rate of 2.25 B/g that the same starter set-up achieved in the Access to Air and its Effect on Yeast Growth in Starters experiment. The measured growth rate for the starter with Fermcap S was slightly lower but should be seen as equal to the other high stir speed starter due to the uncertainty in cell counting.
Conclusion
Stir plate stir speed and resulting vortex development does have an impact on yeast growth. The obvious conclusion is that one should choose a larger flask if that leads to a better vortex. Given that yeast growth depends on the amount of available extract an interesting trade-off becomes apparent: Is it better to have a smaller volume of higher gravity starter with a good vortex or a larger volume of lower gravity starter with a lesser vortex. I’ll be investigating this trade-off soon.
Opinion
I’m not a fan of foam control agents like Fermcap S in brewing. While it can be argued that it is safe (although the manufacturer recommends removal through filtration) it represents a shortcut that home brewers should not embrace. Especially the ones that are complaining about the shortcuts that large commercial brewers are taking. I’m including it in my research in simply because I want to see if the foam produced by a starter gets in the way of the yeast’s access to oxygen. In this case it did not make a difference which was likely due to the fact that the starters did not develop much foam to begin with and the vortex was quite strong.


Cool! Not surprising because you see this all the time in shake-flask and spin-flask cultures, but cool nonetheless to show this in homebrew setting. Power of knowledge!
Yes, more growth with more stirring/shaking makes sense. But home brewers won’t believe you unless there is data.
I’m seeing some interesting results in the fermentation tests (25 Plato wort) for the yeast grown during the aeration experiment. According to weight loss during fermentation the yeast grown with airlock and foil cover is ahead of the other ones. What the heck. Too early to draw any conclusions. I still have to count the cells for their actual pitching rate and take a hydrometer reading. The latter won’t happen for another week. There will be a more official post on that for sure.
Yeah, common sense and homebrewers don’t always get along. I’ve seen people write that what I do is wrong because hemocytometer is not as good as sediment volume for calculating amount of yeast in a starter.
What exactly do you mean by “According to weight loss during fermentation the yeast grown with airlock and foil cover is ahead of the other ones.”
When CO2 escapes the weight of the beer drops and one can detect this difference using a scale. I have done this here to keep track of the fermentation progress. It’s not straight forward to calculate the sg from the weight loss due to supersaturation and the CO2 in the head space but it’s good enough to see which of the samples is fermenting faster than the others. In this case the yeasts gown with air lock and foil cover are ahead.
OK, with airlock and foil cover grow faster as compared to what? To the samples that are completely sealed?
I’ll have to refer you to the post I haven’t written yet. That will clear up any confusion.
Nice write up and pics. Here’s to spinning up the stir plates! An increase of 33-66% is a nice bonus for spinning a little faster.
Would the starter be grown faster too? I’d love to see a comparison to non-stirred starters so we can plug that into the yeast calculator.
I’m not sure if faster spinning also causes the yeast to ferment the starter faster. I think it is best to allow each starter stage about 48 hrs if you want to decant the starter beer. Yeast in an unfinished starter doesn’t like to settle.
As for the still starters, I plan to experiment with that as well, but I think that folks serious about starters will eventually get themselves a stir plate.
Kai, thanks for sharing all your research. I have always wondered if there is a need to keep the stir plate running until reaching final gravity is needed for maximum yeast growth. I assume your research is with running through to final gravity, have you looked into the effects of this verse stopping at different times in the ferment process?
Kai. Thanks for all your diligent work. I agree, it’s funny how homebrewer’s want proof, yet if you have a certain name they take anything you say as proof.
I’ve tried to link and use your data numerous times in homebrewtalk and eventually got a bit tired with that community and left.
The human mind and personality are a very strange thing indeed.
Thanks again.
Hi Kai,
I sincerely appreciate the amount of time and effort you’ve contributed to this thorough experiment. I’m new to the stir plate application and would appreciate your response to my inquiry. The experiment proves a larger volume of yeast created by the vigorous, circulating vortex. Here’s my neophyte question: Is this large volume entirely healthy as I would think that the swift speed of the stir bar would create some sort of damage to the cell wall. Sure they would bud but are they creating healthy “sister” cells? Do yeast get damaged by strong vortex conditions? I’m not being a wise guy, just wondering what you think about this part of the process. More yeast are created but are they in some stage of the petant mutant? When you’ve pitched the larger starters do they attenuate as you expect? Thanks for thinking about my “Q’s” and any insight you can lend.
Keep up the interesting and informative work!
Best regards,
Steve
I don’t think that the speeds at which we brewers stir our starters do damage to the cells. At least I have not seen this being reported.
Any idea what sort of RPMs were you running and how large was the vortex?
I’m wondering if there is a point where it becomes counterproductive or has no increased benefit. I just finished building an epic atirplate and am wondering how fast to puah it. Thanks!
I don’t know the RPMs since my stir plates don’t show them. I only have pics of what the starter looked like. At some point there is little change and going faster won’t do much more.
Any data on rpm and growth rate?
Ive got myself a digital stirplate.
http://www.digitalhomebrew.com/digital-stirplate-v3
Was hoping to get an idea what rpm your “yeast calc” stir-plate calculation was calculated at. Then ill have a better idea how much growth im getting.
tx
Mine is made by Pioneer and has 33 1/2, 45 and 78 rpm
Great work kai!
I got myself a couple of digital stirplates http://www.digitalhomebrew.com/digital-stirplate-v3
Would like an idea as to what your speeds were so I can get an accurate growth calculation. But I suppose your description is pretty good.
Keep up the work. Your resurch is invaluable to Brewing!
Sorry Kai, But these findings seem to contradict Steves findings here…
http://www.woodlandbrew.com/2015/01/cell-density-meter-update.html?m=1
What’s your take on this?
You drew me out of hiding with this one
I don’t think Steve’s experiment is valid for the conclusions he drew. He is comparing a 3ml still fermentation in a vial with a ~300 ml stirred fermentation in a flask. To draw conclusions about agitation vs. no agitation you need to compare the same wort amount in the same size and shape vessel. I also don’t follow his approach to cell density measurement. If he is measuring the light absorption in the vial w/o shaking it, the cell density at the end of fermentation would drop since cells are settling to the bottom. If he is shaking them up to evenly distribute them for counting he doesn’t have a still starter.
Kai
Kai, I also realized the batch size difference.
But I think Steve was building on an assumption from previous experimentation.
I diddnt initially see the drawback in measurement, you have a point. Id be interested to see how Steve sees this.
Kai,
While I agree, that Steve’s comparisons are in-congruent and therefore not probably significant, your confidence intervals overlap in all but the slow group. Kudos for calculating and including them as most people would not have but I don’t think you can comment on any differences except slow vs. the rest. You would need repeat experimentation to narrow those intervals.
The mass measurements are probably better than the absorption spec that Steve was using but oxygen fixation for use in building a cell wall (the whole reason we think aeration is good) would actually increase the mass in the flask while fermentation decreased it. Fermentation rate is probably representative of the cell counts but a better way to measure the yeast would be concentration under a microscope. Not that everyone has one of those.
Thanks for gathering the data though, it’s probably the best low tech representation of what’s actually going on.
The results of these experiments should largely be evaluated on a qualitative basis. Exact formulas for growth rates are difficult to establish since they seems to depend on many parameters. Most of which are difficult to measure or control for the home brewer. While this particular experiment does not disprove that one can get the same or better growth in a still starter (no experiment can prove anything) it supports a trend that I have seen in other experiments as well.
Steve mentions that the Crabtree Effect prevents oxygen uptake. This is misunderstanding of the Crabtree Effect. That effect is all about the presence of anaerobic metabolism once there is a sufficient amount of sugar available to the yeast. It does not say there won’t be any aerobic metabolism. I’m touching on that in my NHC presentation on the topic of yeast growth.
I don’t see details specifying the time duration of this experiment. Were all starters allowed to run to completion, or were they measured after 48 hours? If they were not finished fermenting, I imagine growth would continue beyond the point they were measured at.
I just reread the article and I’m surprised that I did not note how long they were allowed to ferment. I remember letting all of them ferment until the last one showed signs of fermentation (usually the absence of foam).
Here’s a video about how the mechanical action of stirring can measurably heat your starter (https://www.youtube.com/watch?v=GsmvvigROt4) so I wonder if the increased growth can be attributed to the warmer temperature of the vigorously stirred starter more so than the agitation itself?
That’s an interesting thought. I assumed that all starters would be at the same temperature. But even if the temperature was different the difference was likely not big enough to be responsible for the increased growth. However, to completely eliminate this effect, the experiments would have to be repeated.
So for a given amount of food yeast can only grow to the amount that they have food for? T or F
Stirring affects time not quantity. T or F