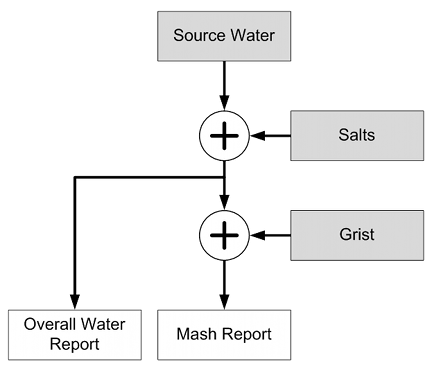
After I conducted the Access to Air and its Effect on Yeast Growth in Starters experiment I also started fermentation tests with that yeast. The tests were designed to stress the yeast during a high gravity wort fermentation that would show differences in the yeast’s fermentation performance. However, it did not show a clear correlation between the yeast’s access to air during propagation and its fermentation performance. It did however show a difference in final yeast viability which suggests that stressing the yeast with an even higher gravity fermentation could show a correlation between yeast propagation and fermentation performance.
Methods and Materials
1 day after the yeast propagation was complete the yeast had settled and the supernatant starter beer was decanted leaving behind a lose slurry of yeast. 3 ml of this yeast slurry was taken from each flask and added to amounts of 25 Plato wort (ranging from 276 to 280 g) in pint sized Mason Jars. The wort was unhopped and prepared from dried malt extract and water. After being boiled for 10 min it was allowed to cool and was then filtered to remove hot break and most cold break.
Yeast was mixed into the wort using a stir plate and initial pitch rate was assessed through cell counts. The jar was then covered with a lid but not closed tightly to allow CO2 to escape. Each jar was shaken vigorously to aerate it. All of the fermentation experiments were weighed on a regular basis to keep track of the fermentation progress. Fermentation happened in a 20 C ambient temperature environment.
After 15 days of fermentation the apparent extract content was assessed using a hydrometer (0.990 – 1.020 range) and thermometer for temperature correction. The total cell count was determined through cell counts and viability was assessed with methylene blue (1 drop of 1% w/w methylene blue to a 5 ml yeast suspension).
Results and Discussion
The following table shows fermentation conditions and statistics that were collected for all 4 fermentations.
| Tag |
A |
B |
C |
D |
|
| yeast prop |
airlock |
foil |
air injected |
no cover |
|
| initial extract |
25 |
Plato |
| temperature |
20 |
C |
| fermentation time |
15 |
days |
| initial pitch rate |
42 |
49 |
65 |
47 |
B/L |
| highest fermentation speed* |
3.9 |
6.4 |
4.5 |
3.9 |
Plato/day |
| final pH |
4.76 |
4.76 |
4.67 |
4.67 |
|
| final extract |
5 |
4.9 |
5.1 |
5.4 |
Plato |
| ADF |
80.0% |
80.4% |
79.6% |
78.4% |
|
| specific growth |
0.19 |
0.29 |
0.11 |
0.14 |
B/g |
| final viability (Metylene Blue) |
60% |
52% |
81% |
91% |
|
*fermentation speed was determined from the highest rate of weight loss.
Due to the inconsistent yeast slurry densities initial pitch rate was not sufficiently controlled by pitching measured amounts of yeast slurry. This resulted in pitching rates ranging from 42 through 65 Billion per liter. But these different pitching rates do not show a correlation to either the fermentation speed, final attenuation or yeast growth.
The attenuation limit of the wort was not assessed but the resulting attenuation levels are fairly close to each other and other experiments with this dried malt extract suggest an attenuation limit of around 80% ADF. Since all yeasts got close to this attenuation limit subsequent experiments should increase the initial extract to 30 Plato to increase yeast stress factors during fermentation. This will make the achieved attenuation level a better indication of the yeast’s ability to cope with these stress factors.
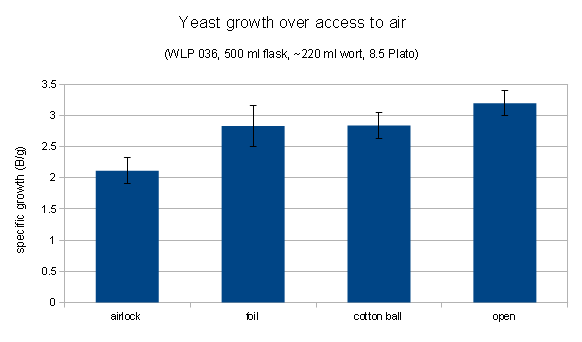
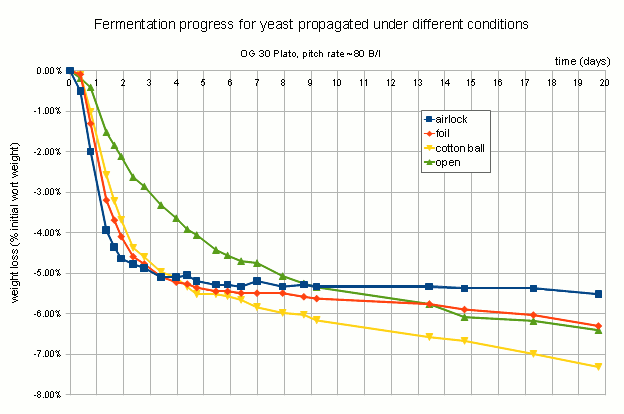
It was expected that the yeast that had the most access to oxygen during propagation, which was C, would show the best fermentation performance. That could not be observed in this experiment. In fact, yeast grown with less access to air (airlock and foil covered), showed better attenuation and fermentation speed.
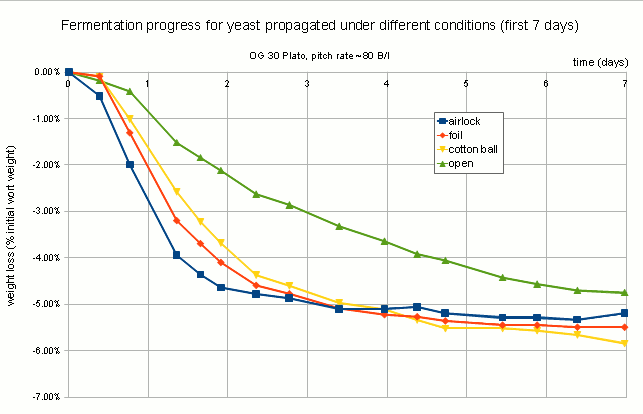
The fermentation performance was assessed indirectly through a measurement of escaped CO2 based on the loss of weight during fermentation. Given the rather large outlier for A (air lock covered yeast propagation), which finished with about 1% less weight than the other fermentations while the actual attenuation were fairly similar between all fermentations, these results need to be treated with caution. If this holds true in subsequent experiments it would be an indication that less aerobically grown yeast (less O2) is better prepared for fermentation and thus does a better job initially than yeast grown more aerobically.
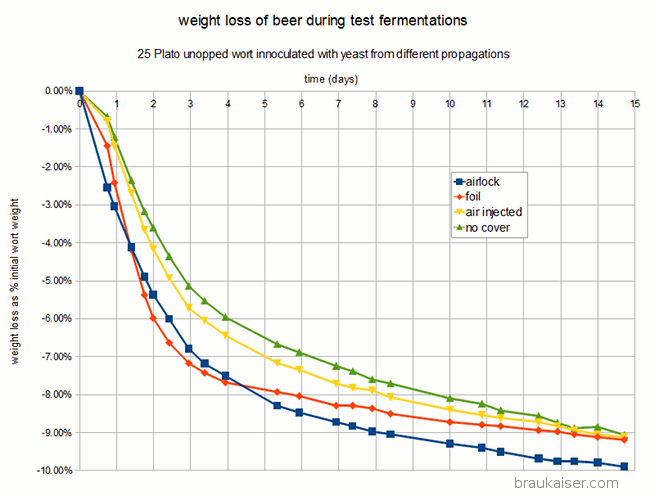
CO2 escape during fermentation plotted as % initial weight loss over time. Fermentation was done in loosely lidded Mason Jars which may have allowed for uneven gas exchange. All 4 samples finished at roughly the same final extract level and larger overall weight loss of “airlock” should be seen as an outlier. Experiments need to be repeated in airlocked fermentation vessels. Note that the labels refer to how the yeast was propagated and not how the test fermentations were conducted.
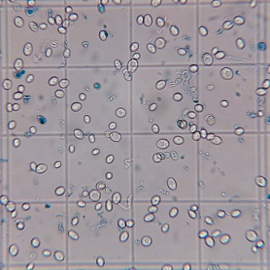
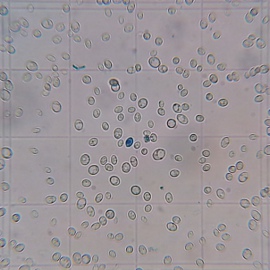
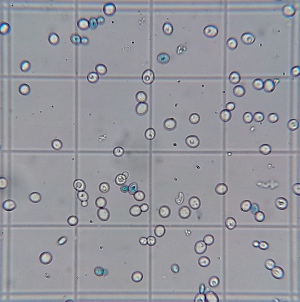
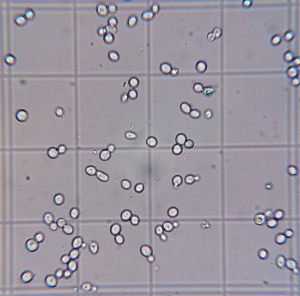
A stark difference was notable in the viabilities of the yeast sediment. Yeast grown with more access to air (air injected and open) showed significantly more viable cells after 15 days of fermentation and a final alcohol concentration of about 10.5% v/v. This suggests that those yeasts may do better during the late phase of fermentation and that they could have outperformed the differently propagated yeasts in higher original gravity worts. The most straightforward explanation of the better health is that those yeasts had larger sterol reserves which they shared with their offsprings and which better protected them from the toxic alcohol environment. Methylene Blue is known to overestimate viability and these experiments should be redone with a stain that is known to be more reliable (Trypan Blue) or plate counts.
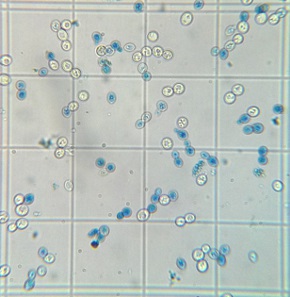 Final cell culture of wort fermented with yeast propagated in a foil covered flask. Stained with Methylene Blue. Stained blue cells are presumed to be dead cells. |
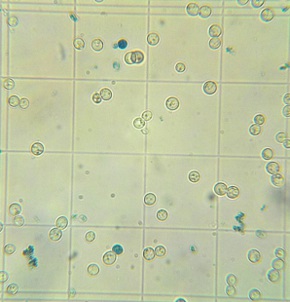 Final cell culture of wort fermented with yeast propagated in a flask open to the air. Stained with Methylene Blue. Stained blue cells are presumed to be dead cells. |
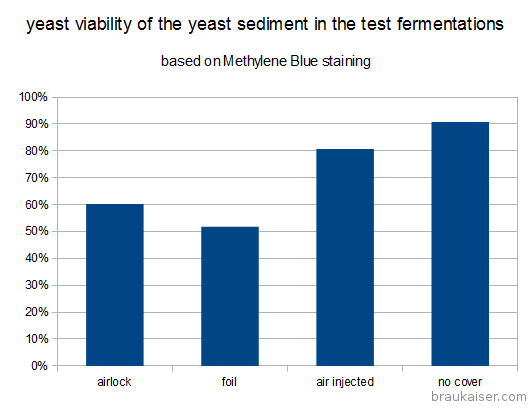
Tested viability (Methylene Blue) of the yeast sediment after 15 days fermentation of a 25 Plato unhopped wort. The labels refer to the access to air that the pitched yeast had during propagation.
Conclusion
While this experiment was intended to show fermentation performance differences for yeast grown under different propagation conditions it fell short to do so. A repeat of the experiment is needed for more conclusive data. Despite its shortcomings this experiment resulted in data that yeast grown with more access to air is better able to withstand high alcohol environments than yeast grown with limited air access if the initial population size is about the same.
The results also suggest that yeast grown with less air access, presumably less aerobic growth, ferments faster and may lead to better attenuation. More experimentation is needed to confirm this effect.